MedicationAgreement-v2.1(2023EN): verschil tussen versies
Nieuwe pagina aangemaakt met '<!-- Hieronder wordt een transclude page aangeroepen --> {{Versions-2.16.840.1.113883.2.4.3.11.60.40.3.9.6(EN)|1|MedicationAgreement-v2.1(2023EN)}} <!-- Tot hier de transclude page --> ==General information<!--hdGeneralInformation-->== Name<!--hdName-->: '''nl.zorg.MedicationAgreement''' link=Medicatieafspraak-v2.1(2023NL)<BR> Version<!--hdVersion-->: '''2.1''' <br> HCIM Status<!--hdStatus-->:Final<br> Release<!--hdPublication-->: '''...' |
Geen bewerkingssamenvatting |
||
| Regel 54: | Regel 54: | ||
|style="width:250px; "|DCM::PublicationStatus||Prepublished | |style="width:250px; "|DCM::PublicationStatus||Prepublished | ||
|- | |- | ||
|style="width:250px; "|DCM::ReviewerList||Projectgroep Medicatieproces & | |style="width:250px; "|DCM::ReviewerList||Projectgroep Medicatieproces & Architectuurgroep Registratie aan de Bron | ||
|- | |- | ||
|style="width:250px; "|DCM::RevisionDate||17-07-2023 | |style="width:250px; "|DCM::RevisionDate||17-07-2023 | ||
| Regel 92: | Regel 92: | ||
|- | |- | ||
|style="width:75px; "|[[ZIBIssues500_1#ZIB-911 | ZIB-911 ]] | |style="width:75px; "|[[ZIBIssues500_1#ZIB-911 | ZIB-911 ]] | ||
|Correction English | |Correction English "Farmaceutical" | ||
|- | |- | ||
|style="width:75px; "|[[ZIBIssues500_2#ZIB-1020 | ZIB-1020 ]] | |style="width:75px; "|[[ZIBIssues500_2#ZIB-1020 | ZIB-1020 ]] | ||
| Regel 98: | Regel 98: | ||
|- | |- | ||
|style="width:75px; "|[[ZIBIssues500_2#ZIB-1021 | ZIB-1021 ]] | |style="width:75px; "|[[ZIBIssues500_2#ZIB-1021 | ZIB-1021 ]] | ||
|Element | |Element 'Geannuleerd indicator' uit MA verwijderen | ||
|} | |} | ||
| Regel 111: | Regel 111: | ||
|- | |- | ||
|style="width:75px; "|[[ZIBIssues500_2#ZIB-1457 | ZIB-1457 ]] | |style="width:75px; "|[[ZIBIssues500_2#ZIB-1457 | ZIB-1457 ]] | ||
|Verwijderen woord | |Verwijderen woord 'voorlopig' | ||
|- | |- | ||
|style="width:75px; "|[[ZIBIssues500_2#ZIB-1458 | ZIB-1458 ]] | |style="width:75px; "|[[ZIBIssues500_2#ZIB-1458 | ZIB-1458 ]] | ||
|Verwijderen sectie | |Verwijderen sectie 'Instructions' bij MedicationAgreement | ||
|- | |- | ||
|style="width:75px; "|[[ZIBIssues500_3#ZIB-1575 | ZIB-1575 ]] | |style="width:75px; "|[[ZIBIssues500_3#ZIB-1575 | ZIB-1575 ]] | ||
| Regel 127: | Regel 127: | ||
|- | |- | ||
|style="width:75px; "|[[ZIBIssues500_3#ZIB-1685 | ZIB-1685 ]] | |style="width:75px; "|[[ZIBIssues500_3#ZIB-1685 | ZIB-1685 ]] | ||
|Medicatiebouwstenen MA, TA, MGB - engelse vertaling van | |Medicatiebouwstenen MA, TA, MGB - engelse vertaling van 'tot en met' | ||
|- | |- | ||
|style="width:75px; "|[[ZIBIssues500_3#ZIB-1696 | ZIB-1696 ]] | |style="width:75px; "|[[ZIBIssues500_3#ZIB-1696 | ZIB-1696 ]] | ||
|MedicatieafspraakAanvullendeInformatieCodelijst heeft | |MedicatieafspraakAanvullendeInformatieCodelijst heeft 'verkeerde' oid voor codesysteem | ||
|- | |- | ||
|style="width:75px; "|[[ZIBIssues500_3#ZIB-1720 | ZIB-1720 ]] | |style="width:75px; "|[[ZIBIssues500_3#ZIB-1720 | ZIB-1720 ]] | ||
| Regel 270: | Regel 270: | ||
|Start date: This is the time at which the agreement was to take effect (or took effect or will take effect). This is the time at which the instructions for use in this agreement start. In the case of an agreement to discontinue use, this is the start date of the original medication agreement. The end date indicates from when the medication is to be discontinued. | |Start date: This is the time at which the agreement was to take effect (or took effect or will take effect). This is the time at which the instructions for use in this agreement start. In the case of an agreement to discontinue use, this is the start date of the original medication agreement. The end date indicates from when the medication is to be discontinued. | ||
<b>Duration:</b> The intended duration of use. E.g. 5 days or 8 weeks. It is not allowed to indicate the duration in months, because different months have a variable duration in days. | |||
<font color="#212121"><b>End date:</b> The time at which the period of use ends (or ended or will end). In the case of an agreement to discontinue use, this is </font>the time at which the medication is to be discontinued. To avoid confusion between 'to' and 'up until and including', the submission of time is always mandatory for the end date. | |||
With medication for an indefinite period only a start date is indicated. | With medication for an indefinite period only a start date is indicated. | ||
| Regel 440: | Regel 440: | ||
|Bewust hoge dosering | |Bewust hoge dosering | ||
|1 | |1 | ||
|G- | |G-std thesaurus 2050 | ||
|2.16.840.1.113883.2.4.3.11.60.20.77.5.2.14.2050 | |2.16.840.1.113883.2.4.3.11.60.20.77.5.2.14.2050 | ||
|Bewust hoge dosering | |Bewust hoge dosering | ||
| Regel 446: | Regel 446: | ||
|Bewust lage dosering | |Bewust lage dosering | ||
|2 | |2 | ||
|G- | |G-std thesaurus 2050 | ||
|2.16.840.1.113883.2.4.3.11.60.20.77.5.2.14.2050 | |2.16.840.1.113883.2.4.3.11.60.20.77.5.2.14.2050 | ||
|Bewust lage dosering | |Bewust lage dosering | ||
| Regel 452: | Regel 452: | ||
|Bewust afwijkende toedieningsweg | |Bewust afwijkende toedieningsweg | ||
|3 | |3 | ||
|G- | |G-std thesaurus 2050 | ||
|2.16.840.1.113883.2.4.3.11.60.20.77.5.2.14.2050 | |2.16.840.1.113883.2.4.3.11.60.20.77.5.2.14.2050 | ||
|Bewust afwijkende toedieningsweg | |Bewust afwijkende toedieningsweg | ||
| Regel 458: | Regel 458: | ||
|Medische noodzaak | |Medische noodzaak | ||
|4 | |4 | ||
|G- | |G-std thesaurus 2050 | ||
|2.16.840.1.113883.2.4.3.11.60.20.77.5.2.14.2050 | |2.16.840.1.113883.2.4.3.11.60.20.77.5.2.14.2050 | ||
|Medische noodzaak | |Medische noodzaak | ||
| Regel 464: | Regel 464: | ||
|Profylaxe | |Profylaxe | ||
|5 | |5 | ||
|G- | |G-std thesaurus 2050 | ||
|2.16.840.1.113883.2.4.3.11.60.20.77.5.2.14.2050 | |2.16.840.1.113883.2.4.3.11.60.20.77.5.2.14.2050 | ||
|Profylaxe | |Profylaxe | ||
| Regel 470: | Regel 470: | ||
|Wijziging in GDS per direct | |Wijziging in GDS per direct | ||
|6 | |6 | ||
|G- | |G-std thesaurus 2050 | ||
|2.16.840.1.113883.2.4.3.11.60.20.77.5.2.14.2050 | |2.16.840.1.113883.2.4.3.11.60.20.77.5.2.14.2050 | ||
|Wijziging in GDS per direct | |Wijziging in GDS per direct | ||
| Regel 476: | Regel 476: | ||
|Wijziging in GDS per rolwissel | |Wijziging in GDS per rolwissel | ||
|7 | |7 | ||
|G- | |G-std thesaurus 2050 | ||
|2.16.840.1.113883.2.4.3.11.60.20.77.5.2.14.2050 | |2.16.840.1.113883.2.4.3.11.60.20.77.5.2.14.2050 | ||
|Wijziging in GDS per rolwissel | |Wijziging in GDS per rolwissel | ||
| Regel 482: | Regel 482: | ||
|Bewust afwijken van leeftijdsgrenzen | |Bewust afwijken van leeftijdsgrenzen | ||
|8 | |8 | ||
|G- | |G-std thesaurus 2050 | ||
|2.16.840.1.113883.2.4.3.11.60.20.77.5.2.14.2050 | |2.16.840.1.113883.2.4.3.11.60.20.77.5.2.14.2050 | ||
|Bewust afwijken van leeftijdsgrenzen | |Bewust afwijken van leeftijdsgrenzen | ||
| Regel 488: | Regel 488: | ||
|Bewust van te lage nierfunctie voor deze dosering | |Bewust van te lage nierfunctie voor deze dosering | ||
|9 | |9 | ||
|G- | |G-std thesaurus 2050 | ||
|2.16.840.1.113883.2.4.3.11.60.20.77.5.2.14.2050 | |2.16.840.1.113883.2.4.3.11.60.20.77.5.2.14.2050 | ||
|Bewust van te lage nierfunctie voor deze dosering | |Bewust van te lage nierfunctie voor deze dosering | ||
| Regel 494: | Regel 494: | ||
|Bewust van eerder gemelde overgevoeligheid | |Bewust van eerder gemelde overgevoeligheid | ||
|10 | |10 | ||
|G- | |G-std thesaurus 2050 | ||
|2.16.840.1.113883.2.4.3.11.60.20.77.5.2.14.2050 | |2.16.840.1.113883.2.4.3.11.60.20.77.5.2.14.2050 | ||
|Bewust van eerder gemelde overgevoeligheid | |Bewust van eerder gemelde overgevoeligheid | ||
| Regel 618: | Regel 618: | ||
|SNOMED CT | |SNOMED CT | ||
|2.16.840.1.113883.6.96 | |2.16.840.1.113883.6.96 | ||
|Wens | |Wens patiënt [DEPRECATED] | ||
|-style="vertical-align:top; " | |-style="vertical-align:top; " | ||
|<span title = "Behandeling volgens afspraak met | |<span title = "Behandeling volgens afspraak met patiënt (situation)">Patient agreed with treatment</span> | ||
|[https://terminologie.nictiz.nl/art-decor/snomed-ct?conceptId=112211000146102 112211000146102] | |[https://terminologie.nictiz.nl/art-decor/snomed-ct?conceptId=112211000146102 112211000146102] | ||
|SNOMED CT | |SNOMED CT | ||
| Regel 656: | Regel 656: | ||
|Verandering in labwaarde | |Verandering in labwaarde | ||
|-style="vertical-align:top; " | |-style="vertical-align:top; " | ||
|<span title = " | |<span title = "Patiëntvriendelijk-Nederlandse taalreferentieset op leesniveau B1 (foundation metadata concept)">Patient-friendly Dutch at B1 reading level language reference set</span> | ||
|[https://terminologie.nictiz.nl/art-decor/snomed-ct?conceptId=160161000146108 160161000146108] | |[https://terminologie.nictiz.nl/art-decor/snomed-ct?conceptId=160161000146108 160161000146108] | ||
|SNOMED CT | |SNOMED CT | ||
| Regel 662: | Regel 662: | ||
|Dubbele registratie van medicamenteuze behandeling | |Dubbele registratie van medicamenteuze behandeling | ||
|-style="vertical-align:top; " | |-style="vertical-align:top; " | ||
|<span title = "Medicatiegebruik van | |<span title = "Medicatiegebruik van patiënt volgens afspraak (finding)">Patient takes medication as prescribed</span> | ||
|[https://terminologie.nictiz.nl/art-decor/snomed-ct?conceptId=112221000146107 112221000146107] | |[https://terminologie.nictiz.nl/art-decor/snomed-ct?conceptId=112221000146107 112221000146107] | ||
|SNOMED CT | |SNOMED CT | ||
| Regel 690: | Regel 690: | ||
*[[MedicationAdministration2-v2.0.1(2023EN)|MedicationAdministration2-v2.0.1]] | *[[MedicationAdministration2-v2.0.1(2023EN)|MedicationAdministration2-v2.0.1]] | ||
==Technical specifications in HL7v3 CDA and HL7 FHIR<!--ftHeader-->== | ==Technical specifications in HL7v3 CDA and HL7 FHIR<!--ftHeader-->== | ||
To exchange information based on health and care information models, additional, more technical specifications are required. | To exchange information based on health and care information models, additional, more technical specifications are required.<BR> | ||
Not every environment can handle the same technical specifications. For this reason, there are several types of technical specifications:<!--ftReferenceIntro--> | Not every environment can handle the same technical specifications. For this reason, there are several types of technical specifications:<!--ftReferenceIntro--> | ||
<ul> | <ul> | ||
<li> | <li>HL7® version 3 CDA compatible specifications, available through the Nictiz ART-DECOR® environment<!--ftArtDecorReference--> {{ArtDecorLinks|2023|9.6}}</li> | ||
<li> | <li>HL7® FHIR® compatible specifications, available through the Nictiz environment on the Simplifier FHIR<!--ftSimplifierReference--> {{SimplefierLinks|2023|Medicatieafspraak}}</li> | ||
</ul> | </ul> | ||
==Downloads<!--ftDownloadTitle-->== | ==Downloads<!--ftDownloadTitle-->== | ||
| Regel 706: | Regel 706: | ||
</ul> | </ul> | ||
Conditions for use are located on the mainpage<!--ftConditions--> [[Bestand:list2.png|link=HCIM_Mainpage<!--wikiMainpage-->]]<BR> | Conditions for use are located on the mainpage<!--ftConditions--> [[Bestand:list2.png|link=HCIM_Mainpage<!--wikiMainpage-->]]<BR> | ||
This page is generated on | This page is generated on 31/10/2023 18:34:54 with ZibExtraction v. 9.3.8704.31782<!--ftDate--> <BR> | ||
----- | ----- | ||
<div style="text-align: right; direction: ltr; margin-left: 1em;" >[[Bestand: Back 16.png| link= HCIM_Release<!--wikiReleasePage-->_2023(EN)]] [[HCIM_Release<!--wikiReleasePage-->_2023(EN) |Back to HCIM list<!--hdBackToMainPage--> ]]</div> | <div style="text-align: right; direction: ltr; margin-left: 1em;" >[[Bestand: Back 16.png| link= HCIM_Release<!--wikiReleasePage-->_2023(EN)]] [[HCIM_Release<!--wikiReleasePage-->_2023(EN) |Back to HCIM list<!--hdBackToMainPage--> ]]</div> | ||
Huidige versie van 31 okt 2023 om 21:00
General information
Name: nl.zorg.MedicationAgreement ![]()
Version: 2.1
HCIM Status:Final
Release: 2023
Release status: Prepublished
Release date: 15-10-2023
Metadata
| DCM::CoderList | Projectgroep Medicatieproces |
| DCM::ContactInformation.Address | |
| DCM::ContactInformation.Name | * |
| DCM::ContactInformation.Telecom | |
| DCM::ContentAuthorList | Projectgroep Medicatieproces |
| DCM::CreationDate | 1-2-2017 |
| DCM::DeprecatedDate | |
| DCM::DescriptionLanguage | nl |
| DCM::EndorsingAuthority.Address | |
| DCM::EndorsingAuthority.Name | PM |
| DCM::EndorsingAuthority.Telecom | |
| DCM::Id | 2.16.840.1.113883.2.4.3.11.60.40.3.9.6 |
| DCM::KeywordList | Medicatie, Afspraak |
| DCM::LifecycleStatus | Final |
| DCM::ModelerList | Kerngroep Registratie aan de Bron |
| DCM::Name | nl.zorg.Medicatieafspraak |
| DCM::PublicationDate | 15-10-2023 |
| DCM::PublicationStatus | Prepublished |
| DCM::ReviewerList | Projectgroep Medicatieproces & Architectuurgroep Registratie aan de Bron |
| DCM::RevisionDate | 17-07-2023 |
| DCM::Supersedes | nl.zorg.Medicatieafspraak-v2.0 |
| DCM::Version | 2.1 |
| HCIM::PublicationLanguage | EN |
Revision History
Only available in Dutch
Publicatieversie 1.0 (04-09-2017)
Publicatieversie 1.0.1 (31-12-2017)
| ZIB-618 | Hernoemen Verstrekking naar Medicatieverstrekking |
| ZIB-643 | Kleine tekstuele verbeteringen |
Publicatieversie 1.1 (31-01-2020)
| ZIB-888 | terminologie reden voor stoppen medicatie verbeteren en SNOMED coderingen zibs met meditatieproces gelijktrekken |
Publicatieversie 1.2 (01-09-2020)
| ZIB-911 | Correction English "Farmaceutical" |
| ZIB-1020 | Zib Gewicht en Zib Lengte uit Zib Medicatieafspraak halen |
| ZIB-1021 | Element 'Geannuleerd indicator' uit MA verwijderen |
Publicatieversie 1.3 (01-12-2021)
| ZIB-1261 | Medicatieafspraak waardelijst MedicatieafspraakStopTypeCodelijst bevat vervallen SNOMED CT codes |
| ZIB-1454 | Onduidelijkheid codes bij StopType codelijsten |
| ZIB-1457 | Verwijderen woord 'voorlopig' |
| ZIB-1458 | Verwijderen sectie 'Instructions' bij MedicationAgreement |
| ZIB-1575 | Deprecated termen en waardelijsten uit de prepublicatie halen |
Publicatieversie 2.0 (10-06-2022)
| ZIB-1659 | Aanpassen terminologiecodering MA in zibMA |
| ZIB-1685 | Medicatiebouwstenen MA, TA, MGB - engelse vertaling van 'tot en met' |
| ZIB-1696 | MedicatieafspraakAanvullendeInformatieCodelijst heeft 'verkeerde' oid voor codesysteem |
| ZIB-1720 | Verzameling errata bouwsteen Medicatieafspraak |
Publicatieversie 2.1 (15-10-2023)
| ZIB-1852 | In de Engelse versie van Medicatieafspraak heeft de waardenlijst ReasonModificationOrDiscontinuation een foute naam |
| ZIB-1939 | Voorbeeld bij Medicatieafspraak prepublicatie 2022 klopt niet meer |
| ZIB-1940 | RedenWijzigen-StakenCodelijst - SNOMED-term bij 159691000146109 |
Concept
A medication agreement is a prescriber’s proposal for a patient to use medication. An agreement to discontinue the use of medication is also a medication agreement.
Purpose
The goal of the medication agreement is to provide insight into the agreements reached between the prescriber and the patient on the use of medication.
Information Model
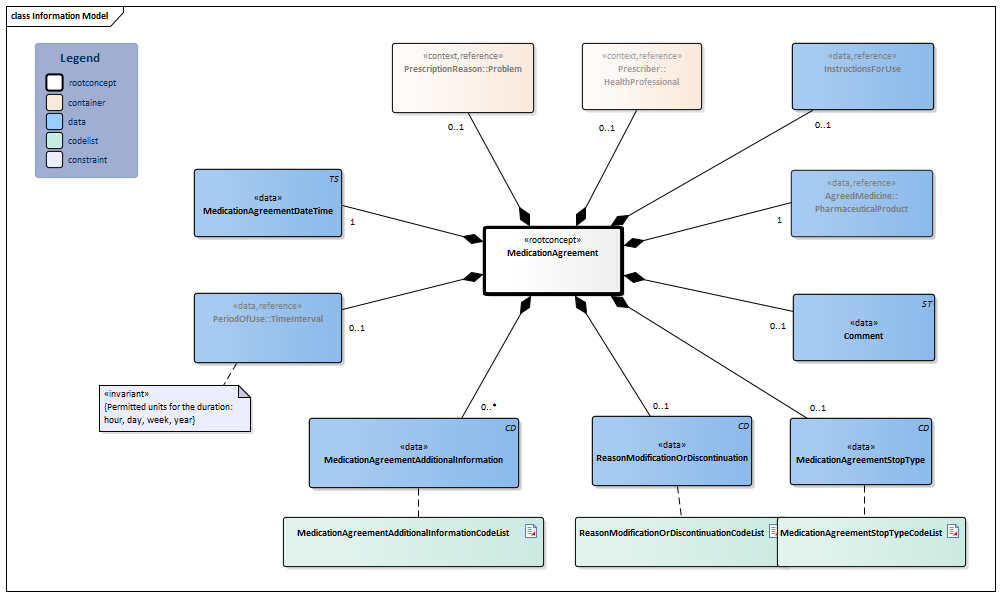
| Type | Id | Concept | Card. | Definition | DefinitionCode | Reference | ||||||||
| NL-CM:9.6.9580 | Root concept of the MedicationAgreement information model. This root concept contains all data elements of the MedicationAgreement information model. |
|
||||||||||||
| NL-CM:9.6.1030 | 0..1 | The health professional that entered the medication agreement with the patient. |
| |||||||||||
| NL-CM:9.6.23133 | 0..1 | The medical reason for the prescription or for use of the medication. This can be used to enter a medical indication which was the direct cause for prescription or for use of the medication in question.
It can concern every type of problem (or condition) of the patient, almost all diagnoses, complaints or symptoms. Please note: The BST401T file of the G standard contains a “special reference” to indicate that “exchange of the reason for prescription is essential”. |
|
| ||||||||||
| NL-CM:9.6.19925 | 1 | The medicine agreed upon to be used. |
| |||||||||||
| NL-CM:9.6.23240 | 0..1 | Instructions for the use of the medication, e.g. dose and route of administration. |
| |||||||||||
| NL-CM:9.6.19757 | 1 | The time at which the agreement was made.
Appointment date + time are required (order of the appointments must be clear in cases with multiple appointments on one day) |
||||||||||||
| NL-CM:9.6.19936 | 0..1 | Start date: This is the time at which the agreement was to take effect (or took effect or will take effect). This is the time at which the instructions for use in this agreement start. In the case of an agreement to discontinue use, this is the start date of the original medication agreement. The end date indicates from when the medication is to be discontinued.
Duration: The intended duration of use. E.g. 5 days or 8 weeks. It is not allowed to indicate the duration in months, because different months have a variable duration in days. End date: The time at which the period of use ends (or ended or will end). In the case of an agreement to discontinue use, this is the time at which the medication is to be discontinued. To avoid confusion between 'to' and 'up until and including', the submission of time is always mandatory for the end date. With medication for an indefinite period only a start date is indicated. |
| |||||||||||
| NL-CM:9.6.19954 | 0..1 | Stop type, the manner in which this medication is discontinued (temporary or definitive). |
|
| ||||||||||
| NL-CM:9.6.22094 | 0..1 | Reason for modification or discontinuation of the medication agreement. |
|
| ||||||||||
| NL-CM:9.6.23283 | 0..* | Additional information includes details on the structure of the agreement made that are relevant for pharmacovigilance and fulfillment by the pharmacist. This can be used e.g. to indicate that there was a conscious decision to deviate from the norm or that the agreement is to be structured in a certain way.
See also the Instructions section for more information about use of the element. |
| |||||||||||
| NL-CM:9.6.22273 | 0..1 | Comments regarding to the medication agreement. For example: in consultation with the medical specialist. |
|
|||||||||||
Columns Concept and DefinitionCode: hover over the values for more information
For explanation of the symbols, please see the legend page ![]()
Example Instances
Only available in Dutch
| MedicatieAfspraak DatumTijd | Gebruiksperiode | Afgesproken geneesmiddel | Voorschrijver | MedicatieAfspraak Stoptype | MedicatieAfspraakRedenWijzigenOfStaken | ||
| Ingangsdatum | Einddatum | Duur | FarmaceutischProduct | Zorgverlener | |||
| Naamgegevens | |||||||
| 18-9-2016 18:00:00 | 08-09-16 | 18-09-16 | Lisinopril tablet 10mg | D. Bakker | Definitief | Geen of onvoldoende effect | |
| 6-3-2016 9:12:30 | 06-03-2016 | Methotrexaat injvlst 25mg/ml 0,6 ml | R. Jansen | ||||
| RedenVan Voorschrijven | Gebruiksinstructie | |||
| Probleem | Omschrijving | Doseerinstructie | ||
| Doseerduur | Dosering |Keerdosis | Toedieningsschema |Frequentie |Interval |Toedientijd |Weekdag |Dagdeel | ||
| Van 8-9-2017 tot 18-9-2017 1x per dag 1 stuk. Vanaf 18-9-2017 staken. | 1 stuk | 1x per dag | ||
| Reumatoïde Artritis | Vanaf 6 maart 2016 1x per week op maandag om 14uur 15 mg (=0,6 ml) | 15 mg (=0,6 ml) | 1x per week op maandag (14u) | |
Valuesets
MedicationAgreementAdditionalInformationCodeList
| Valueset OID: 2.16.840.1.113883.2.4.3.11.60.40.2.9.6.3 | Binding: Extensible |
| Conceptname | Conceptcode | Codesystem name | Codesystem OID | Description |
| Bewust hoge dosering | 1 | G-std thesaurus 2050 | 2.16.840.1.113883.2.4.3.11.60.20.77.5.2.14.2050 | Bewust hoge dosering |
| Bewust lage dosering | 2 | G-std thesaurus 2050 | 2.16.840.1.113883.2.4.3.11.60.20.77.5.2.14.2050 | Bewust lage dosering |
| Bewust afwijkende toedieningsweg | 3 | G-std thesaurus 2050 | 2.16.840.1.113883.2.4.3.11.60.20.77.5.2.14.2050 | Bewust afwijkende toedieningsweg |
| Medische noodzaak | 4 | G-std thesaurus 2050 | 2.16.840.1.113883.2.4.3.11.60.20.77.5.2.14.2050 | Medische noodzaak |
| Profylaxe | 5 | G-std thesaurus 2050 | 2.16.840.1.113883.2.4.3.11.60.20.77.5.2.14.2050 | Profylaxe |
| Wijziging in GDS per direct | 6 | G-std thesaurus 2050 | 2.16.840.1.113883.2.4.3.11.60.20.77.5.2.14.2050 | Wijziging in GDS per direct |
| Wijziging in GDS per rolwissel | 7 | G-std thesaurus 2050 | 2.16.840.1.113883.2.4.3.11.60.20.77.5.2.14.2050 | Wijziging in GDS per rolwissel |
| Bewust afwijken van leeftijdsgrenzen | 8 | G-std thesaurus 2050 | 2.16.840.1.113883.2.4.3.11.60.20.77.5.2.14.2050 | Bewust afwijken van leeftijdsgrenzen |
| Bewust van te lage nierfunctie voor deze dosering | 9 | G-std thesaurus 2050 | 2.16.840.1.113883.2.4.3.11.60.20.77.5.2.14.2050 | Bewust van te lage nierfunctie voor deze dosering |
| Bewust van eerder gemelde overgevoeligheid | 10 | G-std thesaurus 2050 | 2.16.840.1.113883.2.4.3.11.60.20.77.5.2.14.2050 | Bewust van eerder gemelde overgevoeligheid |
MedicationAgreementStopTypeCodeList
| Valueset OID: 2.16.840.1.113883.2.4.3.11.60.40.2.9.6.1 | Binding: Extensible |
| Conceptname | Conceptcode | Codesystem name | Codesystem OID | Description |
| Drug therapy temporarily stopped | 113381000146106 | SNOMED CT | 2.16.840.1.113883.6.96 | Tijdelijke onderbreking van medicamenteuze behandeling (bijvoorbeeld tijdelijk stoppen gebruik vanwege operatie). |
| Drug therapy definitely stopped | 113371000146109 | SNOMED CT | 2.16.840.1.113883.6.96 | Het staken van een bestaande medicamenteuze behandeling. |
ReasonModificationOrDiscontinuationCodeList
| Valueset OID: 2.16.840.1.113883.2.4.3.11.60.40.2.9.6.2 | Binding: Extensible |
| Conceptname | Conceptcode | Codesystem name | Codesystem OID | Description |
| Medication commenced | 266709005 | SNOMED CT | 2.16.840.1.113883.6.96 | Starten medicamenteuze behandeling [DEPRECATED] |
| Administration of medication contraindicated | 438833006 | SNOMED CT | 2.16.840.1.113883.6.96 | Toedienen van medicatie gecontra-indiceerd |
| Medication interaction | 79899007 | SNOMED CT | 2.16.840.1.113883.6.96 | Geneesmiddelinteractie |
| Adverse reaction to drug | 62014003 | SNOMED CT | 2.16.840.1.113883.6.96 | Ongewenste reactie op medicatie en/of drugs |
| Hypersensitivity condition | 473010000 | SNOMED CT | 2.16.840.1.113883.6.96 | Overgevoeligheid [DEPRECATED] |
| Lack of drug action | 58848006 | SNOMED CT | 2.16.840.1.113883.6.96 | Gebrek aan geneesmiddeleffect |
| Drug action too strong | 112251000146103 | SNOMED CT | 2.16.840.1.113883.6.96 | Te sterk effect van medicatie |
| At increased risk of medication side effect | 704417003 | SNOMED CT | 2.16.840.1.113883.6.96 | (Mogelijke) bijwerking [DEPRECATED] |
| Insufficient route of drug administration | 112191000146101 | SNOMED CT | 2.16.840.1.113883.6.96 | Medicatietoedieningsweg voldoet niet |
| Drug treatment not indicated | 183966005 | SNOMED CT | 2.16.840.1.113883.6.96 | Geen indicatie voor medicamenteuze behandeling |
| Medication management changed | 112751000146109 | SNOMED CT | 2.16.840.1.113883.6.96 | Medicatiebeleids veranderd |
| Admission to establishment | 305335007 | SNOMED CT | 2.16.840.1.113883.6.96 | Opname in instelling |
| Patient requested modification of treatment | 159711000146106 | SNOMED CT | 2.16.840.1.113883.6.96 | Verzoek van zorgafnemer om behandeling te wijzigen |
| Patient requested treatment | 184003006 | SNOMED CT | 2.16.840.1.113883.6.96 | Wens patiënt [DEPRECATED] |
| Patient agreed with treatment | 112211000146102 | SNOMED CT | 2.16.840.1.113883.6.96 | Volgens afspraak [DEPRECATED] |
| Treatment previous prescriber continued | 112261000146100 | SNOMED CT | 2.16.840.1.113883.6.96 | Hervatten beleid vorige voorschrijver |
| Planned procedure | 405613005 | SNOMED CT | 2.16.840.1.113883.6.96 | Verrichting gepland |
| Incorrect registration of medication | 159691000146109 | SNOMED CT | 2.16.840.1.113883.6.96 | Foutieve registratie van medicatie |
| Drug not available - out of stock | 182856006 | SNOMED CT | 2.16.840.1.113883.6.96 | Geneesmiddel niet voorradig |
| Change in laboratory test finding | 160131000146104 | SNOMED CT | 2.16.840.1.113883.6.96 | Verandering in labwaarde |
| Patient-friendly Dutch at B1 reading level language reference set | 160161000146108 | SNOMED CT | 2.16.840.1.113883.6.96 | Dubbele registratie van medicamenteuze behandeling |
| Patient takes medication as prescribed | 112221000146107 | SNOMED CT | 2.16.840.1.113883.6.96 | Medicatiegebruik van patient volgens afspraak |
| Overig | OTH | NullFlavor | 2.16.840.1.113883.5.1008 | Overig |
This information model in other releases
- Release 2017, (Version 1.0.1)
- Prerelease 2018-2, (Version 1.0.1)
- Prerelease 2019-2, (Version 1.1)
- Release 2020, (Version 1.2)
- Prerelease 2021-2, (Version 1.3)
- Prerelease 2022-1, (Version 2.0)
- Release 2024, (Version 4.0)
- Prerelease 2026-1, (Version 4.1)
Information model references
This information model refers to
- HealthProfessional-v4.0
- InstructionsForUse-v2.0.1
- PharmaceuticalProduct-v2.2.1
- Problem-v4.6.1
- TimeInterval-v1.2
This information model is used in
Technical specifications in HL7v3 CDA and HL7 FHIR
To exchange information based on health and care information models, additional, more technical specifications are required.
Not every environment can handle the same technical specifications. For this reason, there are several types of technical specifications:
- HL7® version 3 CDA compatible specifications, available through the Nictiz ART-DECOR® environment

- HL7® FHIR® compatible specifications, available through the Nictiz environment on the Simplifier FHIR

Downloads
This information model is also available as pdf file ![]() or as spreadsheet
or as spreadsheet ![]()
About this information
The information in this wikipage is based on Prerelease 2023-1
SNOMED CT and LOINC codes are based on:
- SNOMED Clinical Terms versie: 20230930 [R] (september 2023-editie)
- LOINC version 2.76
Conditions for use are located on the mainpage ![]()
This page is generated on 31/10/2023 18:34:54 with ZibExtraction v. 9.3.8704.31782