PharmaceuticalProduct-v2.0(2017EN): verschil tussen versies
Geen bewerkingssamenvatting |
Geen bewerkingssamenvatting |
||
| (Een tussenliggende versie door dezelfde gebruiker niet weergegeven) | |||
| Regel 1: | Regel 1: | ||
==General information== | <!-- Hieronder wordt een transclude page aangeroepen --> | ||
Name: '''nl.zorg.part.PharmaceuticalProduct''' [[Bestand:NL.png|link=FarmaceutischProduct-v2.0(2017NL)]]<BR> | {{Versions-2.16.840.1.113883.2.4.3.11.60.40.3.9.7(EN)|1|PharmaceuticalProduct-v2.0(2017EN)}} | ||
Version: '''2.0''' <br> | <!-- Tot hier de transclude page --> | ||
HCIM Status:Final<br> | ==General information<!--hdGeneralInformation-->== | ||
Release: '''2017''' <br> | Name<!--hdName-->: '''nl.zorg.part.PharmaceuticalProduct''' [[Bestand:NL.png|link=FarmaceutischProduct-v2.0(2017NL)]]<BR> | ||
Release status: Published<br> | Version<!--hdVersion-->: '''2.0''' <br> | ||
Release date: 31-12-2017 | HCIM Status<!--hdStatus-->:Final<br> | ||
Release<!--hdPublication-->: '''2017''' <br> | |||
Release status<!--hdPublicationStatus-->: Published<br> | |||
Release date<!--hdPublicationDate-->: 31-12-2017 | |||
----- | ----- | ||
<div style="text-align: right; direction: ltr; margin-left: 1em;" >[[Bestand: Back 16.png| link= | <div style="text-align: right; direction: ltr; margin-left: 1em;" >[[Bestand: Back 16.png| link= HCIM_Release<!--wikiReleasePage-->_2017(EN)]] [[HCIM_Release<!--wikiReleasePage-->_2017(EN) |Back to HCIM list<!--hdBackToMainPage--> ]]</div> | ||
==Metadata== | ==Metadata== | ||
| Regel 61: | Regel 64: | ||
==Revision History== | ==Revision History== | ||
<div class="mw-collapsible mw-collapsed"> | <div class="mw-collapsible mw-collapsed"> | ||
''Only available in Dutch'' | ''Only available in Dutch<!--noTranslation-->'' | ||
<u>Publicatieversie 1.0</u> (04-09-2017) | <u>Publicatieversie 1.0</u> (04-09-2017) | ||
| Regel 95: | Regel 98: | ||
==Information Model== | ==Information Model== | ||
<BR> | <BR> | ||
<imagemap> Bestand:PharmaceuticalProduct-v2.0Model( | <imagemap> Bestand:PharmaceuticalProduct-v2.0Model(2017EN).png | center | ||
rect | rect 428 655 539 735 [[#13249]] | ||
rect | rect 569 655 688 735 [[#13260]] | ||
rect | rect 118 828 290 878 [[#IngredientCodeGTINCodelist]] | ||
rect | rect 733 381 909 431 [[#ProductCodeATCCodelist]] | ||
rect | rect 118 760 290 810 [[#IngredientCodeSSKCodelist]] | ||
rect | rect 118 634 290 684 [[#IngredientCodeATCCodelist]] | ||
rect | rect 118 199 295 249 [[#Pharmaceutical formCodelist]] | ||
rect | rect 118 697 290 747 [[#IngredientCodeSNKCodelist]] | ||
rect | rect 118 571 290 621 [[#IngredientCodeZICodelist]] | ||
rect | rect 118 508 290 558 [[#IngredientCodePRKCodelist]] | ||
rect | rect 118 446 290 496 [[#IngredientCodeHPKCodelist]] | ||
rect | rect 115 385 287 435 [[#IngredientCodeGPKCodelist]] | ||
rect | rect 733 311 909 361 [[#ProductCodeZICodelist]] | ||
rect | rect 733 244 909 294 [[#ProductCodeGTINCodelist]] | ||
rect | rect 732 177 908 227 [[#ProductCodePRKCodelist]] | ||
rect | rect 733 110 909 160 [[#ProductCodeHPKCodelist]] | ||
rect | rect 733 43 909 93 [[#ProductCodeGPKCodelist]] | ||
rect | rect 416 284 520 354 [[#13251]] | ||
rect | rect 348 541 438 611 [[#13268]] | ||
rect | rect 470 541 560 611 [[#13273]] | ||
rect | rect 418 416 508 486 [[#13259]] | ||
rect | rect 249 285 362 355 [[#13272]] | ||
rect | rect 579 365 669 435 [[#13253]] | ||
rect | rect 578 170 668 240 [[#13248]] | ||
rect | rect 578 263 668 333 [[#13252]] | ||
rect | rect 421 151 540 221 [[#13255]] | ||
desc none | desc none | ||
</imagemap> | </imagemap> | ||
| Regel 127: | Regel 130: | ||
{| border= "1" width="1500px" style = "font-size: 9.5pt; border: solid 1px silver; border-collapse:collapse;" cellpadding = "3px" cellspacing ="0px" | {| border= "1" width="1500px" style = "font-size: 9.5pt; border: solid 1px silver; border-collapse:collapse;" cellpadding = "3px" cellspacing ="0px" | ||
|-style="background-color: #1F497D; color: white; font-weight: bold; font-variant: small-caps; " | |-style="background-color: #1F497D; color: white; font-weight: bold; font-variant: small-caps; " | ||
|style="width:30px;"|Type||style="width:100px;"|Id||colspan="6 " style="width:140px;"|Concept||Card.||style="width: 600px;"|Definition||style="width:200px;"|DefinitionCode||style="width:200px;"|Reference | |style="width:30px;"|Type<!--imType-->||style="width:100px;"|Id<!--imId-->||colspan="6 " style="width:140px;"|Concept<!--imConcept-->||Card.<!--imCard-->||style="width: 600px;"|Definition<!--imDefinition-->||style="width:200px;"|DefinitionCode<!--imDefinitionCode-->||style="width:200px;"|Reference<!--imReference--> | ||
|-style="vertical-align:top; background-color: #E3E3E3; " | |-style="vertical-align:top; background-color: #E3E3E3; " | ||
|style = "text-align:center" |[[Bestand: block.png| 20px | link=]] | |style = "text-align:center" |[[Bestand: block.png| 20px | link=]] | ||
| Regel 342: | Regel 345: | ||
| | | | ||
|} | |} | ||
Columns Concept and DefinitionCode: hover over the values for more information<BR> | Columns Concept and DefinitionCode: hover over the values for more information<!--imHover--><BR> | ||
For explanation of the symbols, please see the legend page [[Bestand:list2.png|link=Legend]] | For explanation of the symbols, please see the legend page<!--imLegend--> [[Bestand:list2.png|link=Legend<!--wikiLegend-->]] | ||
==Example Instances== | ==Example Instances== | ||
''Only available in Dutch'' | ''Only available in Dutch<!--noTranslation-->'' | ||
{|class="wikitable" width="286px" style= "font-size: 9.5pt;" | {|class="wikitable" width="286px" style= "font-size: 9.5pt;" | ||
| Regel 365: | Regel 368: | ||
{| class="wikitable" width="60%" | {| class="wikitable" width="60%" | ||
|-style='vertical-align:top; background-color: #E8D7BE;' | |-style='vertical-align:top; background-color: #E8D7BE;' | ||
|Valueset OID 2.16.840.1.113883.2.4.3.11.60.40.2.9.7.13 | |Valueset OID<!--vsValuesetOID-->: 2.16.840.1.113883.2.4.3.11.60.40.2.9.7.13 | ||
|Binding<!--vsBindingTag-->: | |||
|} | |} | ||
{| class="wikitable" width="60% " | {| class="wikitable" width="60% " | ||
|-style="background-color: #1F497D; color: white; font-weight: bold; " | |-style="background-color: #1F497D; color: white; font-weight: bold; " | ||
|Conceptname | |Conceptname<!--vsConceptName--> | ||
|Codesystem name||Codesystem OID | |Codesystem name<!--vsConceptSystemName-->||Codesystem OID<!--vsConceptSystemOID--> | ||
|-style="vertical-align:top; " | |-style="vertical-align:top; " | ||
|Alle waarden | |Alle waarden | ||
| Regel 376: | Regel 380: | ||
|2.16.840.1.113883.6.73 | |2.16.840.1.113883.6.73 | ||
|} | |} | ||
=== IngredientCodeGPKCodelist=== | === IngredientCodeGPKCodelist=== | ||
{| class="wikitable" width="60%" | {| class="wikitable" width="60%" | ||
|-style='vertical-align:top; background-color: #E8D7BE;' | |-style='vertical-align:top; background-color: #E8D7BE;' | ||
|Valueset OID 2.16.840.1.113883.2.4.3.11.60.40.2.9.7.9 | |Valueset OID<!--vsValuesetOID-->: 2.16.840.1.113883.2.4.3.11.60.40.2.9.7.9 | ||
|Binding<!--vsBindingTag-->: | |||
|} | |} | ||
{| class="wikitable" width="60% " | {| class="wikitable" width="60% " | ||
|-style="background-color: #1F497D; color: white; font-weight: bold; " | |-style="background-color: #1F497D; color: white; font-weight: bold; " | ||
|Conceptname | |Conceptname<!--vsConceptName--> | ||
|Codesystem name||Codesystem OID | |Codesystem name<!--vsConceptSystemName-->||Codesystem OID<!--vsConceptSystemOID--> | ||
|-style="vertical-align:top; " | |-style="vertical-align:top; " | ||
|Alle waarden | |Alle waarden | ||
| Regel 391: | Regel 395: | ||
|2.16.840.1.113883.2.4.4.1 | |2.16.840.1.113883.2.4.4.1 | ||
|} | |} | ||
=== IngredientCodeGTINCodelist=== | === IngredientCodeGTINCodelist=== | ||
{| class="wikitable" width="60%" | {| class="wikitable" width="60%" | ||
|-style='vertical-align:top; background-color: #E8D7BE;' | |-style='vertical-align:top; background-color: #E8D7BE;' | ||
|Valueset OID 2.16.840.1.113883.2.4.3.11.60.40.2.9.7.16 | |Valueset OID<!--vsValuesetOID-->: 2.16.840.1.113883.2.4.3.11.60.40.2.9.7.16 | ||
|Binding<!--vsBindingTag-->: | |||
|} | |} | ||
{| class="wikitable" width="60% " | {| class="wikitable" width="60% " | ||
|-style="background-color: #1F497D; color: white; font-weight: bold; " | |-style="background-color: #1F497D; color: white; font-weight: bold; " | ||
|Conceptname | |Conceptname<!--vsConceptName--> | ||
|Codesystem name||Codesystem OID | |Codesystem name<!--vsConceptSystemName-->||Codesystem OID<!--vsConceptSystemOID--> | ||
|-style="vertical-align:top; " | |-style="vertical-align:top; " | ||
|Alle waarden | |Alle waarden | ||
| Regel 406: | Regel 410: | ||
|1.3.160 | |1.3.160 | ||
|} | |} | ||
=== IngredientCodeHPKCodelist=== | === IngredientCodeHPKCodelist=== | ||
{| class="wikitable" width="60%" | {| class="wikitable" width="60%" | ||
|-style='vertical-align:top; background-color: #E8D7BE;' | |-style='vertical-align:top; background-color: #E8D7BE;' | ||
|Valueset OID 2.16.840.1.113883.2.4.3.11.60.40.2.9.7.10 | |Valueset OID<!--vsValuesetOID-->: 2.16.840.1.113883.2.4.3.11.60.40.2.9.7.10 | ||
|Binding<!--vsBindingTag-->: | |||
|} | |} | ||
{| class="wikitable" width="60% " | {| class="wikitable" width="60% " | ||
|-style="background-color: #1F497D; color: white; font-weight: bold; " | |-style="background-color: #1F497D; color: white; font-weight: bold; " | ||
|Conceptname | |Conceptname<!--vsConceptName--> | ||
|Codesystem name||Codesystem OID | |Codesystem name<!--vsConceptSystemName-->||Codesystem OID<!--vsConceptSystemOID--> | ||
|-style="vertical-align:top; " | |-style="vertical-align:top; " | ||
|Alle waarden | |Alle waarden | ||
| Regel 421: | Regel 425: | ||
|2.16.840.1.113883.2.4.4.7 | |2.16.840.1.113883.2.4.4.7 | ||
|} | |} | ||
=== IngredientCodePRKCodelist=== | === IngredientCodePRKCodelist=== | ||
{| class="wikitable" width="60%" | {| class="wikitable" width="60%" | ||
|-style='vertical-align:top; background-color: #E8D7BE;' | |-style='vertical-align:top; background-color: #E8D7BE;' | ||
|Valueset OID 2.16.840.1.113883.2.4.3.11.60.40.2.9.7.11 | |Valueset OID<!--vsValuesetOID-->: 2.16.840.1.113883.2.4.3.11.60.40.2.9.7.11 | ||
|Binding<!--vsBindingTag-->: | |||
|} | |} | ||
{| class="wikitable" width="60% " | {| class="wikitable" width="60% " | ||
|-style="background-color: #1F497D; color: white; font-weight: bold; " | |-style="background-color: #1F497D; color: white; font-weight: bold; " | ||
|Conceptname | |Conceptname<!--vsConceptName--> | ||
|Codesystem name||Codesystem OID | |Codesystem name<!--vsConceptSystemName-->||Codesystem OID<!--vsConceptSystemOID--> | ||
|-style="vertical-align:top; " | |-style="vertical-align:top; " | ||
|Alle waarden | |Alle waarden | ||
| Regel 436: | Regel 440: | ||
|2.16.840.1.113883.2.4.4.10 | |2.16.840.1.113883.2.4.4.10 | ||
|} | |} | ||
=== IngredientCodeSNKCodelist=== | === IngredientCodeSNKCodelist=== | ||
{| class="wikitable" width="60%" | {| class="wikitable" width="60%" | ||
|-style='vertical-align:top; background-color: #E8D7BE;' | |-style='vertical-align:top; background-color: #E8D7BE;' | ||
|Valueset OID 2.16.840.1.113883.2.4.3.11.60.40.2.9.7.14 | |Valueset OID<!--vsValuesetOID-->: 2.16.840.1.113883.2.4.3.11.60.40.2.9.7.14 | ||
|Binding<!--vsBindingTag-->: | |||
|} | |} | ||
{| class="wikitable" width="60% " | {| class="wikitable" width="60% " | ||
|-style="background-color: #1F497D; color: white; font-weight: bold; " | |-style="background-color: #1F497D; color: white; font-weight: bold; " | ||
|Conceptname | |Conceptname<!--vsConceptName--> | ||
|Codesystem name||Codesystem OID | |Codesystem name<!--vsConceptSystemName-->||Codesystem OID<!--vsConceptSystemOID--> | ||
|-style="vertical-align:top; " | |-style="vertical-align:top; " | ||
|Alle waarden | |Alle waarden | ||
| Regel 451: | Regel 455: | ||
|2.16.840.1.113883.2.4.4.1.750 | |2.16.840.1.113883.2.4.4.1.750 | ||
|} | |} | ||
=== IngredientCodeSSKCodelist=== | === IngredientCodeSSKCodelist=== | ||
{| class="wikitable" width="60%" | {| class="wikitable" width="60%" | ||
|-style='vertical-align:top; background-color: #E8D7BE;' | |-style='vertical-align:top; background-color: #E8D7BE;' | ||
|Valueset OID 2.16.840.1.113883.2.4.3.11.60.40.2.9.7.15 | |Valueset OID<!--vsValuesetOID-->: 2.16.840.1.113883.2.4.3.11.60.40.2.9.7.15 | ||
|Binding<!--vsBindingTag-->: | |||
|} | |} | ||
{| class="wikitable" width="60% " | {| class="wikitable" width="60% " | ||
|-style="background-color: #1F497D; color: white; font-weight: bold; " | |-style="background-color: #1F497D; color: white; font-weight: bold; " | ||
|Conceptname | |Conceptname<!--vsConceptName--> | ||
|Codesystem name||Codesystem OID | |Codesystem name<!--vsConceptSystemName-->||Codesystem OID<!--vsConceptSystemOID--> | ||
|-style="vertical-align:top; " | |-style="vertical-align:top; " | ||
|Alle waarden | |Alle waarden | ||
| Regel 466: | Regel 470: | ||
|2.16.840.1.113883.2.4.4.1.725 | |2.16.840.1.113883.2.4.4.1.725 | ||
|} | |} | ||
=== IngredientCodeZICodelist=== | === IngredientCodeZICodelist=== | ||
{| class="wikitable" width="60%" | {| class="wikitable" width="60%" | ||
|-style='vertical-align:top; background-color: #E8D7BE;' | |-style='vertical-align:top; background-color: #E8D7BE;' | ||
|Valueset OID 2.16.840.1.113883.2.4.3.11.60.40.2.9.7.12 | |Valueset OID<!--vsValuesetOID-->: 2.16.840.1.113883.2.4.3.11.60.40.2.9.7.12 | ||
|Binding<!--vsBindingTag-->: | |||
|} | |} | ||
{| class="wikitable" width="60% " | {| class="wikitable" width="60% " | ||
|-style="background-color: #1F497D; color: white; font-weight: bold; " | |-style="background-color: #1F497D; color: white; font-weight: bold; " | ||
|Conceptname | |Conceptname<!--vsConceptName--> | ||
|Codesystem name||Codesystem OID | |Codesystem name<!--vsConceptSystemName-->||Codesystem OID<!--vsConceptSystemOID--> | ||
|-style="vertical-align:top; " | |-style="vertical-align:top; " | ||
|Alle waarden | |Alle waarden | ||
| Regel 481: | Regel 485: | ||
|2.16.840.1.113883.2.4.4.8 | |2.16.840.1.113883.2.4.4.8 | ||
|} | |} | ||
=== Pharmaceutical formCodelist=== | === Pharmaceutical formCodelist=== | ||
{| class="wikitable" width="60%" | {| class="wikitable" width="60%" | ||
|-style='vertical-align:top; background-color: #E8D7BE;' | |-style='vertical-align:top; background-color: #E8D7BE;' | ||
|Valueset OID 2.16.840.1.113883.2.4.3.11.60.40.2.9.7.8 | |Valueset OID<!--vsValuesetOID-->: 2.16.840.1.113883.2.4.3.11.60.40.2.9.7.8 | ||
|Binding<!--vsBindingTag-->: | |||
|} | |} | ||
{| class="wikitable" width="60% " | {| class="wikitable" width="60% " | ||
|-style="background-color: #1F497D; color: white; font-weight: bold; " | |-style="background-color: #1F497D; color: white; font-weight: bold; " | ||
|Conceptname | |Conceptname<!--vsConceptName--> | ||
|Codesystem name||Codesystem OID | |Codesystem name<!--vsConceptSystemName-->||Codesystem OID<!--vsConceptSystemOID--> | ||
|-style="vertical-align:top; " | |-style="vertical-align:top; " | ||
|Alle waarden | |Alle waarden | ||
| Regel 496: | Regel 500: | ||
|2.16.840.1.113883.2.4.4.11 | |2.16.840.1.113883.2.4.4.11 | ||
|} | |} | ||
=== ProductCodeATCCodelist=== | === ProductCodeATCCodelist=== | ||
{| class="wikitable" width="60%" | {| class="wikitable" width="60%" | ||
|-style='vertical-align:top; background-color: #E8D7BE;' | |-style='vertical-align:top; background-color: #E8D7BE;' | ||
|Valueset OID 2.16.840.1.113883.2.4.3.11.60.40.2.9.7.7 | |Valueset OID<!--vsValuesetOID-->: 2.16.840.1.113883.2.4.3.11.60.40.2.9.7.7 | ||
|Binding<!--vsBindingTag-->: | |||
|} | |} | ||
{| class="wikitable" width="60% " | {| class="wikitable" width="60% " | ||
|-style="background-color: #1F497D; color: white; font-weight: bold; " | |-style="background-color: #1F497D; color: white; font-weight: bold; " | ||
|Conceptname | |Conceptname<!--vsConceptName--> | ||
|Codesystem name||Codesystem OID | |Codesystem name<!--vsConceptSystemName-->||Codesystem OID<!--vsConceptSystemOID--> | ||
|-style="vertical-align:top; " | |-style="vertical-align:top; " | ||
|Alle waarden | |Alle waarden | ||
| Regel 511: | Regel 515: | ||
|2.16.840.1.113883.6.73 | |2.16.840.1.113883.6.73 | ||
|} | |} | ||
=== ProductCodeGPKCodelist=== | === ProductCodeGPKCodelist=== | ||
{| class="wikitable" width="60%" | {| class="wikitable" width="60%" | ||
|-style='vertical-align:top; background-color: #E8D7BE;' | |-style='vertical-align:top; background-color: #E8D7BE;' | ||
|Valueset OID 2.16.840.1.113883.2.4.3.11.60.40.2.9.7.6 | |Valueset OID<!--vsValuesetOID-->: 2.16.840.1.113883.2.4.3.11.60.40.2.9.7.6 | ||
|Binding<!--vsBindingTag-->: | |||
|} | |} | ||
{| class="wikitable" width="60% " | {| class="wikitable" width="60% " | ||
|-style="background-color: #1F497D; color: white; font-weight: bold; " | |-style="background-color: #1F497D; color: white; font-weight: bold; " | ||
|Conceptname | |Conceptname<!--vsConceptName--> | ||
|Codesystem name||Codesystem OID | |Codesystem name<!--vsConceptSystemName-->||Codesystem OID<!--vsConceptSystemOID--> | ||
|-style="vertical-align:top; " | |-style="vertical-align:top; " | ||
|Alle waarden | |Alle waarden | ||
| Regel 526: | Regel 530: | ||
|2.16.840.1.113883.2.4.4.1 | |2.16.840.1.113883.2.4.4.1 | ||
|} | |} | ||
=== ProductCodeGTINCodelist=== | === ProductCodeGTINCodelist=== | ||
{| class="wikitable" width="60%" | {| class="wikitable" width="60%" | ||
|-style='vertical-align:top; background-color: #E8D7BE;' | |-style='vertical-align:top; background-color: #E8D7BE;' | ||
|Valueset OID 2.16.840.1.113883.2.4.3.11.60.40.2.9.7.2 | |Valueset OID<!--vsValuesetOID-->: 2.16.840.1.113883.2.4.3.11.60.40.2.9.7.2 | ||
|Binding<!--vsBindingTag-->: | |||
|} | |} | ||
{| class="wikitable" width="60% " | {| class="wikitable" width="60% " | ||
|-style="background-color: #1F497D; color: white; font-weight: bold; " | |-style="background-color: #1F497D; color: white; font-weight: bold; " | ||
|Conceptname | |Conceptname<!--vsConceptName--> | ||
|Codesystem name||Codesystem OID | |Codesystem name<!--vsConceptSystemName-->||Codesystem OID<!--vsConceptSystemOID--> | ||
|-style="vertical-align:top; " | |-style="vertical-align:top; " | ||
|Alle waarden | |Alle waarden | ||
| Regel 541: | Regel 545: | ||
|1.3.160 | |1.3.160 | ||
|} | |} | ||
=== ProductCodeHPKCodelist=== | === ProductCodeHPKCodelist=== | ||
{| class="wikitable" width="60%" | {| class="wikitable" width="60%" | ||
|-style='vertical-align:top; background-color: #E8D7BE;' | |-style='vertical-align:top; background-color: #E8D7BE;' | ||
|Valueset OID 2.16.840.1.113883.2.4.3.11.60.40.2.9.7.5 | |Valueset OID<!--vsValuesetOID-->: 2.16.840.1.113883.2.4.3.11.60.40.2.9.7.5 | ||
|Binding<!--vsBindingTag-->: | |||
|} | |} | ||
{| class="wikitable" width="60% " | {| class="wikitable" width="60% " | ||
|-style="background-color: #1F497D; color: white; font-weight: bold; " | |-style="background-color: #1F497D; color: white; font-weight: bold; " | ||
|Conceptname | |Conceptname<!--vsConceptName--> | ||
|Codesystem name||Codesystem OID | |Codesystem name<!--vsConceptSystemName-->||Codesystem OID<!--vsConceptSystemOID--> | ||
|-style="vertical-align:top; " | |-style="vertical-align:top; " | ||
|Alle waarden | |Alle waarden | ||
| Regel 556: | Regel 560: | ||
|2.16.840.1.113883.2.4.4.7 | |2.16.840.1.113883.2.4.4.7 | ||
|} | |} | ||
=== ProductCodePRKCodelist=== | === ProductCodePRKCodelist=== | ||
{| class="wikitable" width="60%" | {| class="wikitable" width="60%" | ||
|-style='vertical-align:top; background-color: #E8D7BE;' | |-style='vertical-align:top; background-color: #E8D7BE;' | ||
|Valueset OID 2.16.840.1.113883.2.4.3.11.60.40.2.9.7.3 | |Valueset OID<!--vsValuesetOID-->: 2.16.840.1.113883.2.4.3.11.60.40.2.9.7.3 | ||
|Binding<!--vsBindingTag-->: | |||
|} | |} | ||
{| class="wikitable" width="60% " | {| class="wikitable" width="60% " | ||
|-style="background-color: #1F497D; color: white; font-weight: bold; " | |-style="background-color: #1F497D; color: white; font-weight: bold; " | ||
|Conceptname | |Conceptname<!--vsConceptName--> | ||
|Codesystem name||Codesystem OID | |Codesystem name<!--vsConceptSystemName-->||Codesystem OID<!--vsConceptSystemOID--> | ||
|-style="vertical-align:top; " | |-style="vertical-align:top; " | ||
|Alle waarden | |Alle waarden | ||
| Regel 571: | Regel 575: | ||
|2.16.840.1.113883.2.4.4.10 | |2.16.840.1.113883.2.4.4.10 | ||
|} | |} | ||
=== ProductCodeZICodelist=== | === ProductCodeZICodelist=== | ||
{| class="wikitable" width="60%" | {| class="wikitable" width="60%" | ||
|-style='vertical-align:top; background-color: #E8D7BE;' | |-style='vertical-align:top; background-color: #E8D7BE;' | ||
|Valueset OID 2.16.840.1.113883.2.4.3.11.60.40.2.9.7.1 | |Valueset OID<!--vsValuesetOID-->: 2.16.840.1.113883.2.4.3.11.60.40.2.9.7.1 | ||
|Binding<!--vsBindingTag-->: | |||
|} | |} | ||
{| class="wikitable" width="60% " | {| class="wikitable" width="60% " | ||
|-style="background-color: #1F497D; color: white; font-weight: bold; " | |-style="background-color: #1F497D; color: white; font-weight: bold; " | ||
|Conceptname | |Conceptname<!--vsConceptName--> | ||
|Codesystem name||Codesystem OID | |Codesystem name<!--vsConceptSystemName-->||Codesystem OID<!--vsConceptSystemOID--> | ||
|-style="vertical-align:top; " | |-style="vertical-align:top; " | ||
|Alle waarden | |Alle waarden | ||
| Regel 587: | Regel 591: | ||
|} | |} | ||
==This information model in other releases<!--ftOtherReleases-->== | |||
==This information model in other releases== | <!-- Hieronder wordt een transclude page aangeroepen --> | ||
--- | {{Versions-2.16.840.1.113883.2.4.3.11.60.40.3.9.7(EN)|2|2017}} | ||
== | <!-- Tot hier de transclude page --> | ||
==Information model references<!--ftReferences-->== | |||
====This information model refers to<!--ftRefersTo-->==== | |||
:-- | |||
====This information model is used in<!--ftReferredBy-->==== | |||
*[[AdministrationAgreement-v1.0.1(2017EN)|AdministrationAgreement-v1.0.1]] | |||
*[[DispenseRequest-v1.0.1(2017EN)|DispenseRequest-v1.0.1]] | |||
*[[MedicationAdministration2-v1.0.1(2017EN)|MedicationAdministration2-v1.0.1]] | |||
*[[MedicationAgreement-v1.0.1(2017EN)|MedicationAgreement-v1.0.1]] | |||
*[[MedicationDispense-v2.0(2017EN)|MedicationDispense-v2.0]] | |||
*[[MedicationUse2-v1.0.1(2017EN)|MedicationUse2-v1.0.1]] | |||
==Technical specifications in HL7v3 CDA and HL7 FHIR<!--ftHeader-->== | |||
To exchange information based on health and care information models, additional, more technical specifications are required.<BR> | To exchange information based on health and care information models, additional, more technical specifications are required.<BR> | ||
Not every environment can handle the same technical specifications. For this reason, there are several types of technical specifications: | Not every environment can handle the same technical specifications. For this reason, there are several types of technical specifications:<!--ftReferenceIntro--> | ||
<ul> | <ul> | ||
<li>HL7® version 3 CDA compatible specifications, available through the Nictiz ART-DECOR® environment [http://decor.nictiz.nl/art-decor/decor-scenarios--zib2017bbr-?id=2.16.840.1.113883.2.4.3.11.60.7.4.2.9.7&effectiveDate=2017-12-31T00:00:00&language=en-US&scenariotree=false [[File:artdecor.jpg|16px|link=]]]</li> | <li>HL7® version 3 CDA compatible specifications, available through the Nictiz ART-DECOR® environment<!--ftArtDecorReference--> [http://decor.nictiz.nl/art-decor/decor-scenarios--zib2017bbr-?id=2.16.840.1.113883.2.4.3.11.60.7.4.2.9.7&effectiveDate=2017-12-31T00:00:00&language=en-US&scenariotree=false [[File:artdecor.jpg|16px|link=]]]</li> | ||
<li>HL7® FHIR® compatible specifications, available through the Nictiz environment on the Simplifier FHIR | <li>HL7® FHIR® compatible specifications, available through the Nictiz environment on the Simplifier FHIR<!--ftSimplifierReference--> {{SimplefierLinks|2017}}</li> | ||
</ul> | </ul> | ||
This information model is also available as [[Media:nl.zorg.part.PharmaceuticalProduct-v2.0(2017EN).pdf|pdf file]] [[File:PDF.png|link=]] or as [[Media:nl.zorg.part.PharmaceuticalProduct-v2.0(2017EN).xlsx|spreadsheet]] [[File:xlsx.png|link=]] | ==Downloads<!--ftDownloadTitle-->== | ||
==About this information== | This information model is also available as [[Media:nl.zorg.part.PharmaceuticalProduct-v2.0(2017EN).pdf|pdf file]] [[File:PDF.png|link=]] or as [[Media:nl.zorg.part.PharmaceuticalProduct-v2.0(2017EN).xlsx|spreadsheet]] [[File:xlsx.png|link=]]<!--ftDownloads--> | ||
The information in this wikipage is based on Release 2017 <BR> | ==About this information<!--ftHeader2-->== | ||
Conditions for use are located on the mainpage [[Bestand:list2.png|link=HCIM_Mainpage]]<BR> | The information in this wikipage is based on<!--ftInfoBase--> Release 2017 <BR> | ||
This page is generated on | Conditions for use are located on the mainpage<!--ftConditions--> [[Bestand:list2.png|link=HCIM_Mainpage<!--wikiMainpage-->]]<BR> | ||
This page is generated on 21/12/2018 14:06:02 with ZibExtraction v. 3.0.6929.24609<!--ftDate--> <BR> | |||
----- | ----- | ||
<div style="text-align: right; direction: ltr; margin-left: 1em;" >[[Bestand: Back 16.png| link= | <div style="text-align: right; direction: ltr; margin-left: 1em;" >[[Bestand: Back 16.png| link= HCIM_Release<!--wikiReleasePage-->_2017(EN)]] [[HCIM_Release<!--wikiReleasePage-->_2017(EN) |Back to HCIM list<!--hdBackToMainPage--> ]]</div> | ||
Huidige versie van 24 dec 2018 om 12:04
General information
Name: nl.zorg.part.PharmaceuticalProduct ![]()
Version: 2.0
HCIM Status:Final
Release: 2017
Release status: Published
Release date: 31-12-2017
Metadata
| DCM::CoderList | Projectgroep Medicatieproces |
| DCM::ContactInformation.Address | |
| DCM::ContactInformation.Name | * |
| DCM::ContactInformation.Telecom | |
| DCM::ContentAuthorList | Projectgroep Medicatieproces |
| DCM::CreationDate | 1-3-2017 |
| DCM::DeprecatedDate | |
| DCM::DescriptionLanguage | nl |
| DCM::EndorsingAuthority.Address | |
| DCM::EndorsingAuthority.Name | PM |
| DCM::EndorsingAuthority.Telecom | |
| DCM::Id | 2.16.840.1.113883.2.4.3.11.60.40.3.9.7 |
| DCM::KeywordList | FarmaceutischProduct |
| DCM::LifecycleStatus | Final |
| DCM::ModelerList | Architectuurgroep Registratie aan de Bron |
| DCM::Name | nl.zorg.part.FarmaceutischProduct |
| DCM::PublicationDate | 31-12-2017 |
| DCM::PublicationStatus | Published |
| DCM::ReviewerList | Projectgroep Medicatieproces & Architectuurgroep Registratie aan de Bron |
| DCM::RevisionDate | 31-12-2017 |
| DCM::Superseeds | nl.zorg.part.Product-v1.0 |
| DCM::Version | 2.0 |
| HCIM::PublicationLanguage | EN |
Revision History
Only available in Dutch
Publicatieversie 1.0 (04-09-2017)
Publicatieversie 2.0 (31-12-2017)
| ZIB-472 | Terminologiekoppeling klopt niet met beschrijving |
| ZIB-618 | Hernoemen Verstrekking naar Medicatieverstrekking |
Concept
The prescribed substance is usually medication. However, medical aids and bandages can also be prescribed and supplied via the pharmacy. Food and blood products do not strictly belong in the medication category, but can be prescribed and supplied by a pharmacy as well.
A type of medication can be indicated with a single code. That code can be chosen from several possible coding systems (concretely: GPK, PRK, HPK or article numbers). Correct use of these codes in the software systems will sufficiently record the composition of the product used, making a complete product specification unnecessary.
In addition to a primary code, alternative codes from other coding systems can also be entered (so that the GPK can be sent along in the event that the patient was registered based on PRK, for example).
Entering multiple ingredients will enable you to display a compound product. If one of the composite parts is liquid, the dosage will be given in milliliters; otherwise it will be given in ‘units’.
In that case, the composition of the medication can be specified implicitly (with the use of a medication code) or explicitly, for example by listing the (active) ingredient(s) of the medication.
Magistral prescriptions can be entered as well. This can be done by means of the option listed above to enter coded ingredients and/or by entering the composition and preparation method as free text.
This is a partial information model
Purpose
The purpose of Product is to unambiguously describe the medication to be used.
Information Model
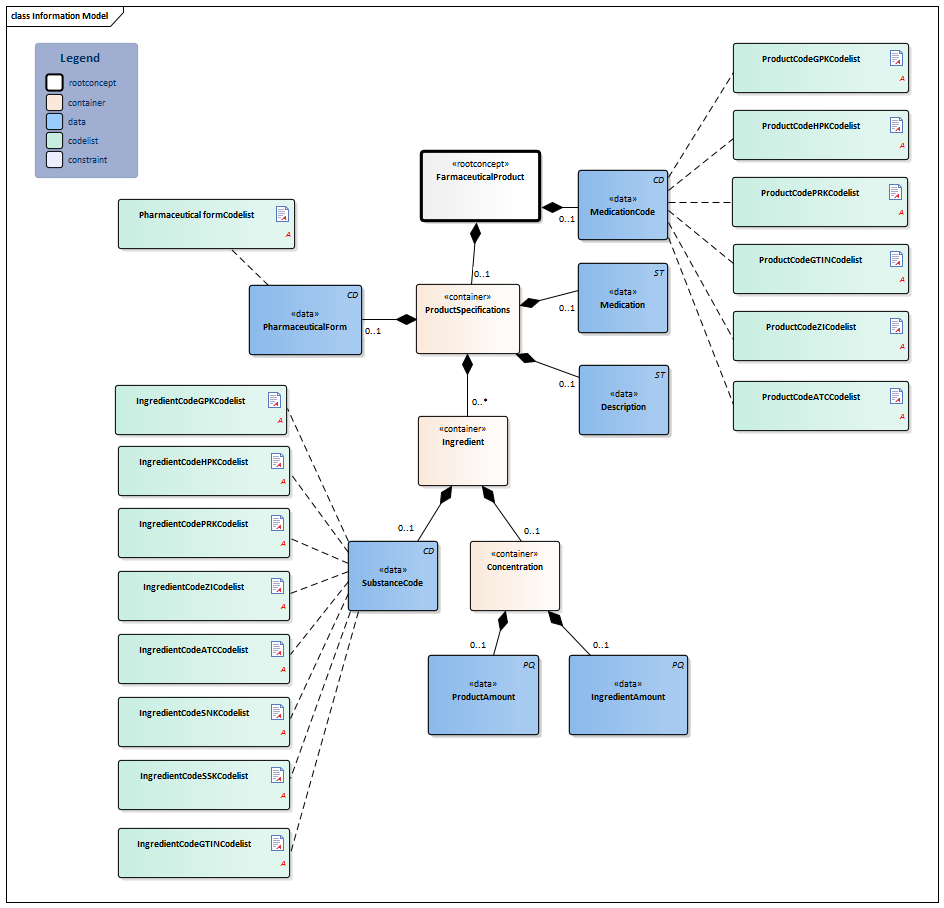
| Type | Id | Concept | Card. | Definition | DefinitionCode | Reference | |||||||||||||||||
| NL-CM:9.7.19926 | Root concept of the FarmaceuticalProduct partial information model. This root concept contains all data elements of the FarmaceuticalProduct partial information model.
The prescribed product is usually a medicine. However, medical aids and bandages can also be prescribed and supplied via the pharmacy. Strictly speaking, food and blood products do not belong in the medication category, but can be prescribed and supplied by a pharmacy as well. A type of medication can be indicated with a single code. That code can be chosen from several possible coding systems (concretely: GPK, PRK, HPK or article numbers). Correct use of these codes in the software systems will sufficiently record the composition of the product used, making a complete product specification unnecessary. In addition to a primary code, alternative codes from other coding systems can also be entered (so that the GPK can be sent along in the event that the patient was registered based on PRK, for example). Entering multiple ingredients will enable you to display a compound product. If one of the composite parts is liquid, the dosage will be given in milliliters; otherwise it will be given in ‘units’. In that case, the composition of the medication can be specified implicitly (with the use of a medication code) or explicitly, for example by listing the (active) substance(s) of the medication. Prescriptions to be prepared by the pharmacy can be entered as well. This can be done by means of the option listed above to enter coded ingredients and/or by entering the composition and preparation method as free text. |
||||||||||||||||||||||
| NL-CM:9.7.19928 | 0..1 | Container of the ProductSpecifications concept. This container contains all data elements of the ProductSpecifications concept.
Product specifications are required if the product code is not sufficient to ascertain the active substances and strength. |
|||||||||||||||||||||
| NL-CM:9.7.19931 | 0..1 | The pharmaceutical form indicates the form of the medication in accordance with the route of administration. Examples include: tablet, suppository, infusion liquid, ointment. If the product has a generic code in the G standard, the form will be known in the G standard. For products without a code (free text, preparation by the pharmacy), the means of administration can be entered. |
| ||||||||||||||||||||
| NL-CM:9.7.19927 | 0..1 | Coding medication in the Netherlands is done on the basis of the G standard (issued by Z-index), which is filled under the direction of KNMP.
The coded medication can be expressed as:
The GTIN enables identification of the product including its origin with a barcode. The ATKODE is the number with which wholesalers link the article to pharmacy systems (e.g. a box with 3 strips of 10 tablets). The HPK is the code for the trade product (with the brand name) as used per dose/per time the medication is taken (1 pill, 1 puff, 1ml) The PRK codes for the same product as the HPK does, but is not linked to a manufacturer (no brand name, no characteristics such as color, geometrical shape etc.). This code will enable a generic prescription, while still defining which trade product can be taken (e.g. a 200ml bag). The generic product code defines the composition of a product, and is sufficient for recording the prescription, but not the order. The prescription code (PRK) was developed and added to the older generic (GPK) and supplier-specific (HPK, ATKODE) coding to enable a generic product to be entered without listing a specific brand on the one hand, and to enable providing enough information to support the pharmacy supplying it on the other. The Substance Name Code (SNK) and the Substance Name Code, in combination with Route of Administration (SSK) are used to prescribe at a more generic level. The GTIN coding is used for the implementation of a barcode scanning standard and to be able to trace the origin of the product. The 90.000.000 number is used in accordance with national agreements. |
| ||||||||||||||||||||
| NL-CM:9.7.19929 | 0..1 | There is no code for medication entered in free text. In these cases, enter the complete description. | |||||||||||||||||||||
| NL-CM:9.7.19784 | 0..1 | A textual description of the type of medication (including relevant properties of the composition and preparation method if possible), which is only used if no coded indication from the G Standard is available (magistral preparation). | |||||||||||||||||||||
| NL-CM:9.7.19932 | 0..* | Container of the Ingredient concept. This container contains all data elements of the Ingredient concept.
A product contains one or more active substances and excipients. These are usually determined by the product code. For medication prepared or compounded by the local pharmacy, each ingredient must be entered separately. The active substances play an important role, as they: a) determine the pharmacotherapeutic effect of the medication and b) serve as the basis for the indication of the strength of the medication (e.g. 200mg). |
|||||||||||||||||||||
| NL-CM:9.7.19934 | 0..1 | Active substance or excipient.
Here, the same codes can be used as for the ProductCode (for dilutions and compounds in particular), but now, the ATC, SSK and SNK codes can also be used to indicate a substance (to list ingredients of local products prepared by the pharmacy).
The ATC is an international classification of pharmaceutical substances without a reference to specific products on the market. Therefore, the ATC code of a generic product will not contain a reference to a certain dose, pharmaceutical form or route of administration; it will only contain a reference to the ingredients (not the amount/concentration/strength). |
|||||||||||||||||||||
| NL-CM:9.7.19933 | 0..1 | The relative amount of this ingredient in this product.
Calculation of Concentration = Ingredient Amount ÷ Product Amount. This could be a concentration if the medication is dissolved in liquid, for example. |
|||||||||||||||||||||
| NL-CM:9.7.22277 | 0..1 | The amount of this ingredient. This is the numerator for the calculation of the concentration. The unit should be selected from the G-Standard (Table 902). | |||||||||||||||||||||
| NL-CM:9.7.22278 | 0..1 | Amount of the product. This is the denominator for the calculation of the concentration.Optionally a translation to NHG table Gebruiksvoorschriften(Table 25) is also allowed. | |||||||||||||||||||||
Columns Concept and DefinitionCode: hover over the values for more information
For explanation of the symbols, please see the legend page ![]()
Example Instances
Only available in Dutch
| Afgesproken geneesmiddel |
| Farmaceutischproduct |
| Lisinopril tablet 10mg |
| Methotrexaat injvlst 25mg/ml 0,6 ml |
Valuesets
IngredientCodeATCCodelist
| Valueset OID: 2.16.840.1.113883.2.4.3.11.60.40.2.9.7.13 | Binding: |
| Conceptname | Codesystem name | Codesystem OID |
| Alle waarden | Anatomic Therapeutic Classification (ATC) | 2.16.840.1.113883.6.73 |
IngredientCodeGPKCodelist
| Valueset OID: 2.16.840.1.113883.2.4.3.11.60.40.2.9.7.9 | Binding: |
| Conceptname | Codesystem name | Codesystem OID |
| Alle waarden | G-Standaard Generieke Product Kode (GPK) | 2.16.840.1.113883.2.4.4.1 |
IngredientCodeGTINCodelist
| Valueset OID: 2.16.840.1.113883.2.4.3.11.60.40.2.9.7.16 | Binding: |
| Conceptname | Codesystem name | Codesystem OID |
| Alle waarden | Global Trade Item Number (GTIN) | 1.3.160 |
IngredientCodeHPKCodelist
| Valueset OID: 2.16.840.1.113883.2.4.3.11.60.40.2.9.7.10 | Binding: |
| Conceptname | Codesystem name | Codesystem OID |
| Alle waarden | G-Standaard Handels Product Kode (HPK) | 2.16.840.1.113883.2.4.4.7 |
IngredientCodePRKCodelist
| Valueset OID: 2.16.840.1.113883.2.4.3.11.60.40.2.9.7.11 | Binding: |
| Conceptname | Codesystem name | Codesystem OID |
| Alle waarden | G-Standaard Voorschrijfproducten (PRK) | 2.16.840.1.113883.2.4.4.10 |
IngredientCodeSNKCodelist
| Valueset OID: 2.16.840.1.113883.2.4.3.11.60.40.2.9.7.14 | Binding: |
| Conceptname | Codesystem name | Codesystem OID |
| Alle waarden | G-standaard Stofnaamcode (SNK) | 2.16.840.1.113883.2.4.4.1.750 |
IngredientCodeSSKCodelist
| Valueset OID: 2.16.840.1.113883.2.4.3.11.60.40.2.9.7.15 | Binding: |
| Conceptname | Codesystem name | Codesystem OID |
| Alle waarden | G-standaard Stofnaamcode i.c.m. toedieningsweg (SSK) | 2.16.840.1.113883.2.4.4.1.725 |
IngredientCodeZICodelist
| Valueset OID: 2.16.840.1.113883.2.4.3.11.60.40.2.9.7.12 | Binding: |
| Conceptname | Codesystem name | Codesystem OID |
| Alle waarden | G-Standaard Artikelen (ook KNMP-nummer, ATKODE) | 2.16.840.1.113883.2.4.4.8 |
Pharmaceutical formCodelist
| Valueset OID: 2.16.840.1.113883.2.4.3.11.60.40.2.9.7.8 | Binding: |
| Conceptname | Codesystem name | Codesystem OID |
| Alle waarden | G-Standaard Farmaceutische vormen | 2.16.840.1.113883.2.4.4.11 |
ProductCodeATCCodelist
| Valueset OID: 2.16.840.1.113883.2.4.3.11.60.40.2.9.7.7 | Binding: |
| Conceptname | Codesystem name | Codesystem OID |
| Alle waarden | Anatomic Therapeutic Classification (ATC) | 2.16.840.1.113883.6.73 |
ProductCodeGPKCodelist
| Valueset OID: 2.16.840.1.113883.2.4.3.11.60.40.2.9.7.6 | Binding: |
| Conceptname | Codesystem name | Codesystem OID |
| Alle waarden | G-Standaard Generieke Product Kode (GPK) | 2.16.840.1.113883.2.4.4.1 |
ProductCodeGTINCodelist
| Valueset OID: 2.16.840.1.113883.2.4.3.11.60.40.2.9.7.2 | Binding: |
| Conceptname | Codesystem name | Codesystem OID |
| Alle waarden | Global Trade Item Number (GTIN) | 1.3.160 |
ProductCodeHPKCodelist
| Valueset OID: 2.16.840.1.113883.2.4.3.11.60.40.2.9.7.5 | Binding: |
| Conceptname | Codesystem name | Codesystem OID |
| Alle waarden | G-Standaard Handels Product Kode (HPK) | 2.16.840.1.113883.2.4.4.7 |
ProductCodePRKCodelist
| Valueset OID: 2.16.840.1.113883.2.4.3.11.60.40.2.9.7.3 | Binding: |
| Conceptname | Codesystem name | Codesystem OID |
| Alle waarden | G-Standaard Voorschrijfproducten (PRK) | 2.16.840.1.113883.2.4.4.10 |
ProductCodeZICodelist
| Valueset OID: 2.16.840.1.113883.2.4.3.11.60.40.2.9.7.1 | Binding: |
| Conceptname | Codesystem name | Codesystem OID |
| Alle waarden | G-Standaard Artikelen (ook KNMP-nummer, ATKODE) | 2.16.840.1.113883.2.4.4.8 |
This information model in other releases
- Prerelease 2018-2, (Version 2.0)
- Prerelease 2019-2, (Version 2.1.1)
- Release 2020, (Version 2.1.2)
- Prerelease 2021-2, (Version 2.1.3)
- Prerelease 2022-1, (Version 2.2)
- Prerelease 2023-1, (Version 2.2.1)
- Prerelease 2024-1, (Versie 2.2.1)
- Release 2024, (Version 2.3)
Information model references
This information model refers to
- --
This information model is used in
- AdministrationAgreement-v1.0.1
- DispenseRequest-v1.0.1
- MedicationAdministration2-v1.0.1
- MedicationAgreement-v1.0.1
- MedicationDispense-v2.0
- MedicationUse2-v1.0.1
Technical specifications in HL7v3 CDA and HL7 FHIR
To exchange information based on health and care information models, additional, more technical specifications are required.
Not every environment can handle the same technical specifications. For this reason, there are several types of technical specifications:
- HL7® version 3 CDA compatible specifications, available through the Nictiz ART-DECOR® environment

- HL7® FHIR® compatible specifications, available through the Nictiz environment on the Simplifier FHIR

Downloads
This information model is also available as pdf file ![]() or as spreadsheet
or as spreadsheet ![]()
About this information
The information in this wikipage is based on Release 2017
Conditions for use are located on the mainpage ![]()
This page is generated on 21/12/2018 14:06:02 with ZibExtraction v. 3.0.6929.24609